Pandemic
Preparedness
The COVID-19 pandemic won't be the last outbreak your hospital will have to face.
HALYARD* supplies PPE from our own Americas-based manufacturing facilities to help you be prepared for what's next.
Now is the time to update your strategic PPE plan—when you can see, and measure—what your facility needs to be ready.
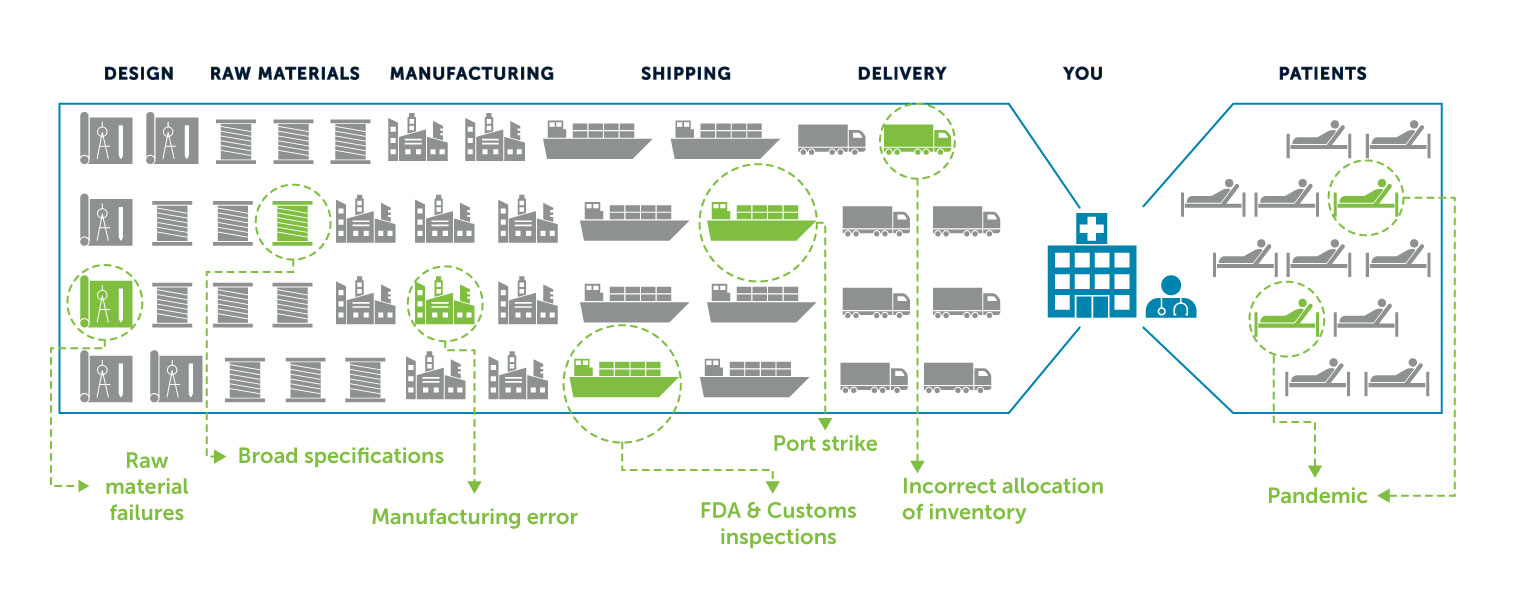
HIV-AIDS, H1N1 flu, Ebola, and now COVID-19 have all highlighted the critical challenges created by global disease outbreaks, including:
- Lack of public-private sector coordination
- Severe warehousing distribution capacity limitations
- A far-flung global supply chain
- Access constraints caused by airport and port restrictions and border closures.
A complex and remote supply chain can break in many places. That’s why “made here” matters when it comes to pandemic planning.
You can count on HALYARD* to help meet your needs during a pandemic event or infectious disease outbreak.
We invest in proximate production capacity and work with our raw material suppliers and distributor partners to meet current and future increased demand, with limited interruptions to our customers.
10 Questions To Ask Before You Stockpile
10 Questions To Ask Before You Stockpile
The questions posed in this downloadable guide are designed to help you begin creating a stockpiling system that will accomplish your pandemic preparedness goals. Whether the infectious outbreak risk is a seasonal flu, pandemic influenza, or a unique pathogen, we can help you determine the type and quantities of PPE needed for your hospital’s unique pathogen scenarios.
NEED HELP ESTIMATING STOCKPILE QUANTITIES?
Contact a HALYARD* representative to help you estimate PPE requirements for pandemic events
Managing in a Post-COVID 19 World
On a recent podcast with the Journal of Healthcare Contracting, experts from Owens & Minor as well as Tampa General Hospital discussed the supply chain and demand challenges hospitals are facing as elective surgeries come back online.
The PPE gear you need for pandemic preparedness
HALYARD* offers quality PPE that meets and often exceeds industry standards for protection from pathogens and fluids to keep healthcare workers safe.
FLUIDSHIELD* Surgical N95 Respirator Mask
100% of HALYARD FLUIDSHIELD N95 Respirators are a medical device—unlike many N95s or KN95s that have received a temporary Emergency Use Authorization for the COVID-19 pandemic. All HALYARD N95s are Made in the Americas, unlike suppliers who rely on supply from Asia. Our duckbill breathing chamber is more than twice as large as the leading competitive surgical N95, and exceeds NIOSH standards for breathability.
- Fluid resistance: 160 mmHg
- Type: Respirator
- SO SOFT* Lining
HALYARD* Isolation Gowns
HALYARD* BASICS* Tri-Layer AAMI2 Over-The-Head Isolation Gown
Provides barrier protection based on AAMI guidelines and helps reduce the risk of contamination and transmission of infectious organisms that lead to HAIs.
- Meets AAMI Level 2 Requirements
- Medium weight 3-layer SMS fabric
- Over-the-head design, tie waist
- Thumb hooks and elastic cuffs
- Ultrasonic Seam Closure
- Closed back
Additional Products to ensure Pandemic safety
Qualitative Fit Test Kits
HALYARD* Face Shield
Request Consultation
Ready to get prepared? Contact one of our representatives today.